|
|||||||||||
|
|||||||||||
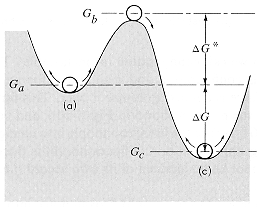
| The
diagram shows a system with a meatstable state (a), an unstable state (b),
and a stable state (c). Energy is required to cause the system to go between
these states.
The "activation energy", DG, for the a -> c process is (Gb - Ga), that for the c -> a process is (Gb - Gc). If
the system is in contact with a thermal reservoir at a temperature T K,
the probability of state (a) acquiring the required activation energy from
thermal fluctuations in the system is given by:
|
|||||||||||
 |
|||||||||||
| From:
Barrett, Nix, and Tetelman,
"The Principles of Engineering Materials," Prentice Hall (1973) |
|||||||||||