From the balanced equations in the the chemistry section, it can be seen that 3 moles of I2 are produced for every mole of IO3- reacted. And each mole of I2 reacts with 2 moles of S2O32-. Therefore 6 moles of S2O32- are required to react with all the I2 produced from 1 mole KIO3-.

The stoichiometric relationship of S2O32- to IO3- is:
![]()
The concentration of Na2S2O3 can be calculated by:
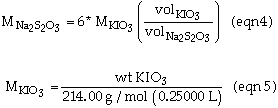
Substitution of eqn 5 into eqn 4 gives eqn 6. This equations allows you to calculate the concentration of Na2S2O3 from experimental numbers.
![]()
where:
Using the rules outlined in part 10 of the lab manual, the upper and lower limits can be calculated by:
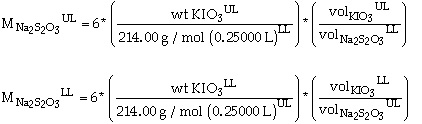
where the uncertainty for:
To determine if two concentrations can be averaged, the concentration from one run must fit between the upper and lower limits of the other run.